Table of Contents
- The Ancient Earth: A World Without Oxygen or Abundant Metals
- Molybdenum’s Biochemical Superpower
- The Great Oxidation Event: A Turning Point for Life and Metals
- Why This Changes Our Understanding of Life’s Origins
- Implications for the Search for Extraterrestrial Life
- Life’s Resilience: A Lesson in Adaptation
The Hidden Metal That Sparked Life: How Molybdenum Fueled Earth’s First Organisms
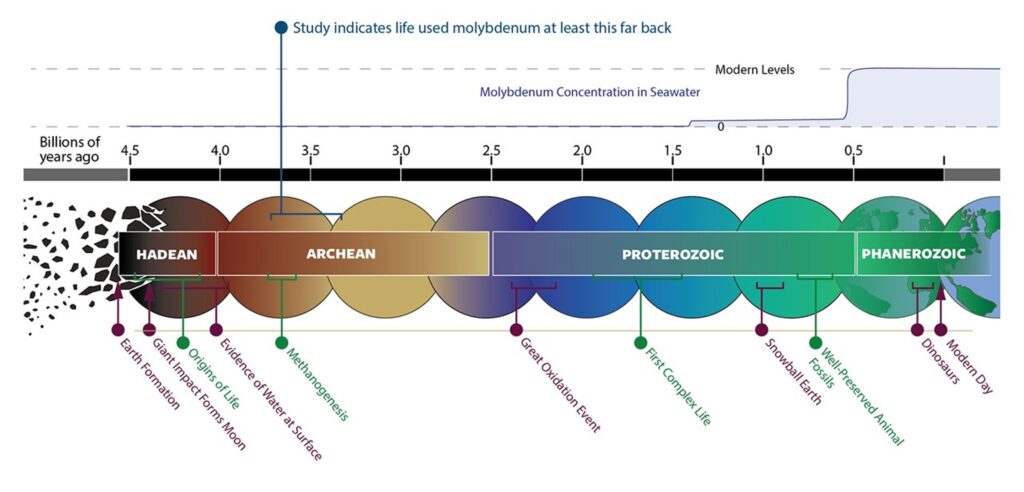
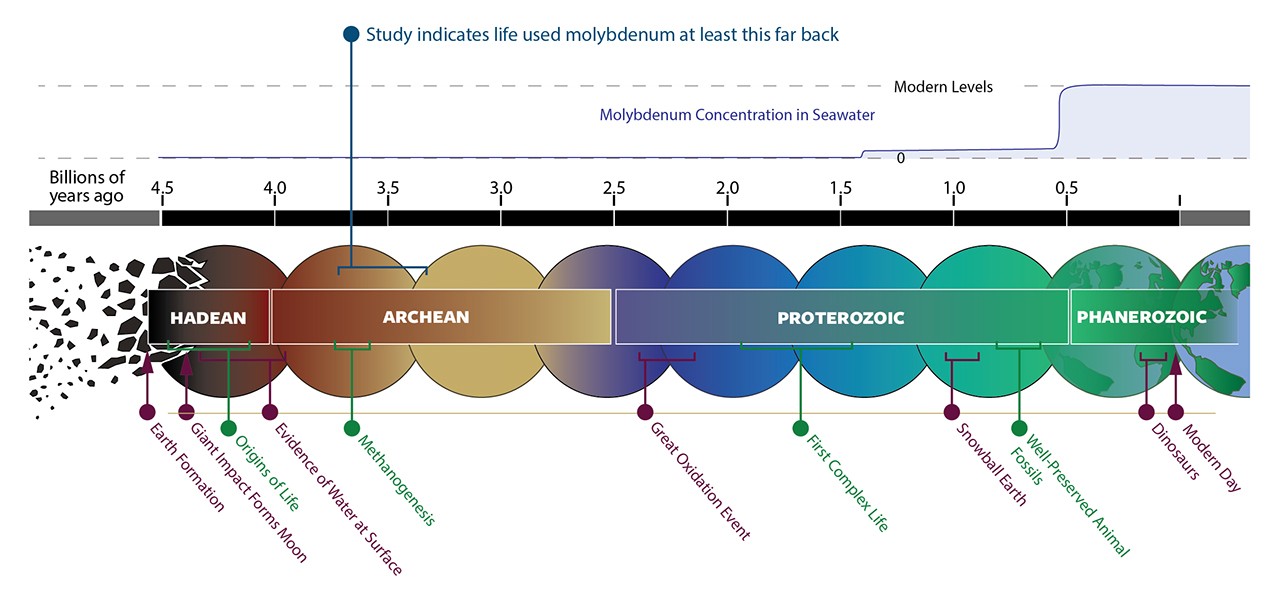
Billions of years before humans walked the Earth, before dinosaurs roamed the continents, and even before oceans fully formed, life was already quietly evolving—powered by a rare metal most people have never heard of. Recent NASA-funded research has uncovered a startling truth: molybdenum, a trace element once nearly absent from Earth’s ancient oceans, played a crucial role in the emergence of early life over 3 billion years ago. This discovery challenges long-held assumptions about how life began and what it needed to survive in a hostile, oxygen-poor world.
For decades, scientists have debated the chemical ingredients that kickstarted life on Earth. While carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur—the so-called “CHNOPS” elements—are well-known building blocks, metals like iron, zinc, and copper have also been recognized as essential cofactors in ancient enzymes. But molybdenum? It was thought to be too scarce to matter in Earth’s primordial soup. Now, a groundbreaking study published in Nature Communications reveals that early life didn’t just survive without abundant molybdenum—it relied on it, even when the metal was vanishingly rare.
This finding rewrites part of the story of life’s origins and offers new clues about how metabolism evolved under extreme environmental constraints. It also has profound implications for the search for life beyond Earth, where similar metal-dependent biochemistry might exist—or might have taken a completely different path.
The Ancient Earth: A World Without Oxygen or Abundant Metals
To understand why molybdenum’s role is so surprising, we must first picture Earth 3.7 billion years ago. Our planet was a volatile, volcanic landscape, still cooling from its formation. The atmosphere lacked free oxygen—a gas that would later become essential for complex life. Instead, it was rich in methane, ammonia, carbon dioxide, and nitrogen, with intense ultraviolet radiation bombarding the surface due to the absence of an ozone layer.
In this harsh environment, the oceans were chemically alien compared to today. Iron was abundant, but many other metals, including molybdenum, were locked away in minerals or present only in trace amounts. Molybdenum, in particular, tends to bind tightly to sulfides and other compounds under low-oxygen conditions, making it biologically inaccessible. This scarcity led scientists to hypothesize that early life might have used alternative metals—like tungsten—to perform vital chemical reactions.
But the new research suggests that life didn’t wait for molybdenum levels to rise. Instead, early organisms evolved sophisticated mechanisms to scavenge and utilize this rare metal efficiently. This implies that molybdenum wasn’t just a convenient tool—it was a necessity for certain metabolic pathways that became foundational to life itself.
Molybdenum’s Biochemical Superpower
So why is molybdenum so special? The answer lies in its unique ability to facilitate electron transfers in chemical reactions—a process central to metabolism. Molybdenum is a key component of a class of enzymes called molybdenum-dependent enzymes, which catalyze some of the most important reactions in biology.
One of the most critical is nitrogen fixation—the conversion of atmospheric nitrogen (N₂) into ammonia (NH₃), a form usable by living organisms. Without this process, life as we know it couldn’t exist, because nitrogen is essential for building proteins and DNA. Today, nitrogen-fixing bacteria use molybdenum-containing enzymes like nitrogenase to perform this miracle. But until now, scientists weren’t sure when this system evolved.
The new study provides strong evidence that molybdenum-based nitrogen fixation was already active over 3 billion years ago. By analyzing ancient rock formations in Australia and South Africa, researchers detected isotopic signatures consistent with biological nitrogen fixation. These signatures align with the presence of molybdenum-dependent enzymes, suggesting that life had already “chosen” molybdenum as its go-to metal for this vital task—even when it was exceedingly rare.
But how did early life manage to acquire enough molybdenum to power these reactions? The answer may lie in localized environments where molybdenum was temporarily concentrated—such as near hydrothermal vents or in shallow tidal pools where evaporation could increase metal concentrations. These microenvironments could have served as “biochemical hotspots” where life first harnessed molybdenum’s power.
The Great Oxidation Event: A Turning Point for Life and Metals
Around 2.45 billion years ago, Earth underwent a dramatic transformation known as the Great Oxidation Event (GOE). Cyanobacteria—early photosynthetic microbes—began producing oxygen as a byproduct of photosynthesis. Over millions of years, oxygen accumulated in the atmosphere and oceans, fundamentally altering Earth’s chemistry.
This shift had a profound effect on metal availability. Oxygen reacts with metals like iron and molybdenum, changing their solubility and reactivity. In particular, molybdenum became much more soluble in oxygen-rich waters, transforming from a rare, locked-up element into a bioavailable resource. Its concentration in the oceans increased by orders of magnitude.
For decades, scientists believed that this rise in molybdenum was a prerequisite for the evolution of complex life. After all, animals and plants rely heavily on molybdenum-dependent enzymes for functions like detoxification and nitrogen metabolism. But the new study turns this idea on its head: life was already using molybdenum long before the GOE.
This means that early organisms didn’t wait for environmental conditions to improve—they adapted to scarcity. They evolved high-affinity transporters to capture molybdenum from dilute solutions and developed enzymes that could function with minimal metal input. In a way, life didn’t just survive in a metal-poor world—it thrived because of its ingenuity.
3.7 billion years ago, levels may have been as low as 1 nanomole per liter—100 times less than today.
Despite this, early life managed to extract and use molybdenum with remarkable efficiency.
Why This Changes Our Understanding of Life’s Origins
The discovery that molybdenum was essential to early life challenges a long-standing hypothesis in astrobiology: the “tungsten world” theory. This idea suggested that life might have started using tungsten—a metal more soluble under anoxic conditions—and only later switched to molybdenum when oxygen levels rose.
But the new evidence shows that molybdenum was already in use billions of years before the Great Oxidation Event. This suggests that life’s metabolic toolkit was more sophisticated than previously thought, and that key biochemical innovations occurred much earlier in Earth’s history.
“Asking when life began using molybdenum is really asking when some of the most consequential metabolic strategies became possible,” said Dr. Betül Kaçar, a co-author of the study and an expert in evolutionary biochemistry. “This metal wasn’t just a passive participant—it was a driver of evolutionary innovation.”
This insight also reshapes our understanding of how life might arise on other planets. If early Earth life could thrive using a rare metal, then perhaps life elsewhere doesn’t need abundant resources to get started. It only needs the right chemistry—and a little ingenuity.
The element molybdenum was first identified in 1778 by Swedish chemist Carl Wilhelm Scheele, who isolated it from the mineral molybdenite. The name comes from the Greek molybdos, meaning “lead,” because the mineral was often confused with lead ore.
Implications for the Search for Extraterrestrial Life
The molybdenum discovery isn’t just a story about Earth’s past—it’s a guide for future exploration. NASA and other space agencies are actively searching for signs of life on Mars, Europa (a moon of Jupiter), and Enceladus (a moon of Saturn). These worlds have environments that may resemble early Earth: cold, oxygen-poor, and rich in volcanic activity.
If life can emerge and persist using scarce metals, then even seemingly inhospitable worlds might harbor microbial ecosystems. Moreover, the presence of molybdenum—or its chemical signatures—in extraterrestrial samples could be a biosignature, indicating past or present biological activity.
Scientists are now developing instruments to detect trace metals in Martian soil and icy plumes from ocean worlds. The ability to identify molybdenum and related elements could help pinpoint the most promising locations for life detection missions.
Humans need about 45 micrograms of molybdenum per day—a tiny amount, but essential for health.
Molybdenum deficiency can lead to neurological issues and impaired growth in animals.
The metal is also used in steel alloys, making it vital for construction and aerospace industries.
Despite its importance, molybdenum ranks only 54th in abundance among elements in Earth’s crust.
Life’s Resilience: A Lesson in Adaptation
At its core, this discovery is a testament to life’s incredible adaptability. Faced with a metal-poor environment, early organisms didn’t give up—they evolved. They developed molecular machines capable of extracting value from scarcity, turning limitation into innovation.
This resilience echoes throughout the history of life on Earth. From surviving mass extinctions to colonizing extreme environments, life has repeatedly found ways to persist against the odds. The story of molybdenum is just one chapter in that ongoing saga—a reminder that the ingredients for life don’t need to be abundant, only essential.
As we look to the stars in search of alien life, we might do well to remember this lesson. Life doesn’t wait for perfect conditions. It emerges where it can, using whatever tools are at hand—even if those tools are as rare as a whisper in the wind.
This article was curated from NASA Research Shows Early Life Relied on Rare Metal via NASA Breaking News
Discover more from GTFyi.com
Subscribe to get the latest posts sent to your email.